The Glow Stack Blend is a single lyophilized vial that co-formulates three of the most-studied repair peptides in modern preclinical literature: GHK-Cu (50 mg), TB-500 (10 mg), and BPC-157 (10 mg). For research workflows that already work with each compound separately, the blend collapses three reconstitution events into one and aligns the three concentration ratios in a fixed stoichiometry. This article walks through what the three components actually do at the bench, how to reconstitute a copper-containing blend without destroying the GHK-Cu chelate, the IU math on a U100 insulin syringe, and where the blend format helps versus where three separate vials remain the better choice. Everything below is framed for in vitro and animal-model research; nothing here is a human dosing recommendation.
1. What is the Glow Stack Blend
A peptide blend is exactly what it sounds like: two or more peptides freeze-dried together inside one sterile vial. The user adds bacteriostatic water once, swirls gently, and draws each subsequent volume from the same multi-dose container. The Glow Stack ships from Bergdorf Bioscience as a research-grade lyophilized blend and pairs the three peptides at a 50 mg / 10 mg / 10 mg ratio for a total of 70 mg of active peptide content per vial.
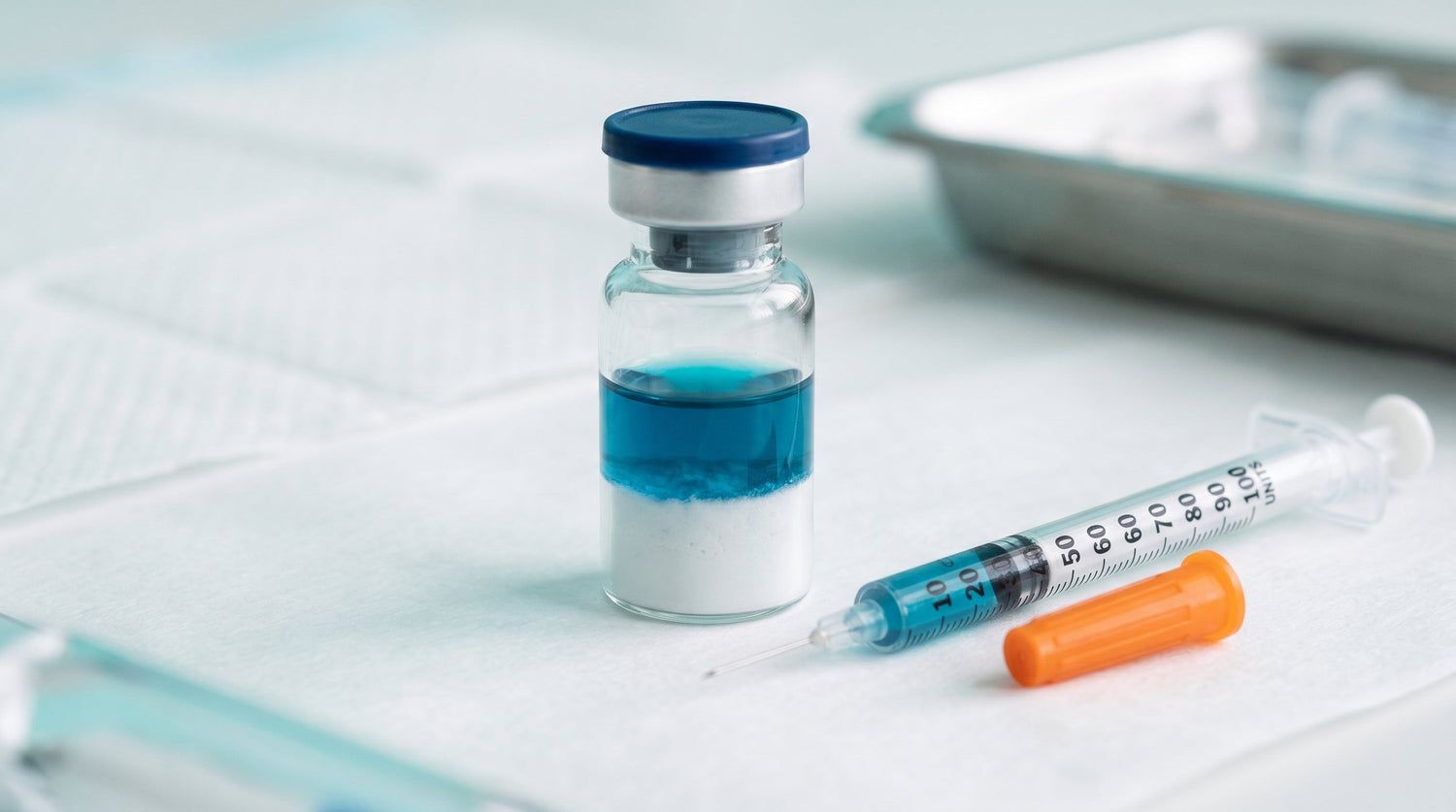
The naming convention reflects the mechanistic story rather than any cosmetic marketing claim: GHK-Cu and TB-500 are widely cited in dermal and connective-tissue repair literature, while BPC-157 brings vascular and gastrointestinal models to the same vial. Each component arrives as a stable lyophilized cake; the deep-blue tint that appears once the vial is reconstituted comes from the GHK-Cu chelate and is the most visible quality indicator of the entire blend.
Composition at a glance
- GHK-Cu: 50 mg per vial. Tripeptide Glycyl-L-Histidyl-L-Lysine bound to a single Cu(II) ion. Theoretical mass 340.79 Da.
- TB-500: 10 mg per vial. Synthetic fragment of Thymosin Beta-4 covering the LKKTETQ active region. Vendor-cited research half-life around 7 hours.
- BPC-157: 10 mg per vial. Pentadecapeptide GEPPPGKPADDAGLV derived from a putative gastric protective sequence. Vendor-cited research half-life around 4 hours.
- Total active peptide: 70 mg.
- Form: White-to-off-white lyophilized powder under inert headspace.
- Stated purity: HPLC purity at or above 99 percent per component, batch-documented in the certificate of analysis.
2. Mechanisms of the three peptides
Each component in the Glow Stack has its own mechanistic literature, and understanding the three pathways separately is the only honest way to think about what the blend can and cannot tell a researcher in a single experimental run.
GHK-Cu: copper transport, ECM remodeling, redox modulation
GHK-Cu is a coordination complex between the tripeptide Gly-His-Lys and Cu(II), first isolated from human plasma by Loren Pickart in 1973. The complex has very high affinity for copper (log K around 16.4) and is widely regarded as a physiological copper transporter. In a 2015 review in BioMed Research International, Pickart, Vasquez-Soltero and Margolina collated Connectivity Map data showing GHK modulates expression of roughly 31 percent of profiled human genes in cultured fibroblasts.
The downstream effects most consistently described in the literature: induction of collagen I and III, elastin, and decorin synthesis (Maquart et al., FEBS Letters, 1988), upregulation of VEGF and basic FGF transcripts, induction of antioxidant defense genes including SOD2 and metallothionein isoforms, and modulation of the MMP-2 and TIMP-1/2 balance toward non-fibrotic remodeling. Hostynek and colleagues quantified percutaneous absorption of the intact GHK-Cu complex through human skin in 2010, supporting bioavailability of the chelate as a unit rather than as separated peptide and copper.
TB-500: actin sequestration and cell migration
TB-500 is the synthetic name commonly applied to the LKKTETQ-containing fragment of Thymosin Beta-4 (Tb4), a 43-amino-acid actin-sequestering peptide present in nearly every mammalian cell type. The active motif binds G-actin monomers and modulates actin polymerization at the leading edge of migrating cells. Bock-Marquette and co-workers reported in Nature (2004) that Tb4 promotes cardiomyocyte survival post-myocardial-infarction in mice through PINCH/ILK/Akt signaling, and Smart and colleagues showed in 2007 that the same peptide mobilizes adult epicardial progenitor cells.
In dermal and corneal models, Tb4 accelerates re-epithelialization (Malinda et al., J Invest Dermatol, 1999; Sosne et al., Exp Eye Res, 2002), suppresses NF-kB-mediated cytokine release, and reduces myofibroblast trans-differentiation, which the authors interpret as an anti-scarring effect. Long-term oncologic safety remains an open research question because the same migration-promoting and pro-angiogenic activity that supports wound closure has, in some murine melanoma models, correlated with increased metastasis.
BPC-157: nitric oxide, angiogenesis, tendon-fibroblast outgrowth
BPC-157 (Body Protection Compound 157) is a 15-amino-acid pentadecapeptide first characterized by the Sikiric group at the University of Zagreb beginning in the early 1990s. The proposed mechanism centers on modulation of the nitric oxide system: BPC-157 counteracts L-NAME-induced eNOS blockade in rat models and supports vascular sprouting through VEGFR2 internalization (Hsieh et al., J Mol Med, 2017). Chang and colleagues showed in 2011 that the peptide upregulates growth-hormone receptor expression on tendon fibroblasts and increases their migration and survival in culture, an effect that has been reproduced across several Achilles- and quadriceps-tendon transection models in rats.
BPC-157 is also notable for an unusual proteolytic stability profile in gastric juice, which forms part of the rationale for oral research applications, although no human pharmacokinetic data have been published. Almost all efficacy data come from a single research group, and independent replication of the larger-effect-size studies remains limited.
3. Synergy hypothesis: a three-axis model
No peer-reviewed study has tested any pairwise combination of GHK-Cu, TB-500, and BPC-157 in vitro or in vivo. The synergy claim that gives the Glow Stack its rationale is therefore mechanistic rather than experimental. The most parsimonious framing is a three-axis model:
- Transcriptional axis (GHK-Cu): upregulates VEGF, bFGF, collagen and elastin transcripts; balances MMP/TIMP for non-fibrotic ECM turnover.
- Cytoskeletal axis (TB-500): sequesters G-actin to enable rapid polymerization at the migrating leading edge; supports endothelial tubulogenesis.
- Receptor and NO axis (BPC-157): drives VEGFR2 internalization, modulates eNOS, supports focal adhesion turnover via FAK and paxillin.
Because each peptide acts on a different node of the same shared pathways (the VEGF axis, NF-kB suppression, ECM remodeling), the combination is theoretically additive rather than redundant. The honest research caveat is that "theoretically additive" is not "experimentally additive" and a researcher who needs to claim synergy must run the combination versus the components in their own model. The blend simply makes that experiment cheaper to set up.
4. Reconstitution of a copper-containing blend
A blend that contains GHK-Cu is not a standard peptide reconstitution. The copper-tripeptide chelate is mechanically and chemically more fragile than either TB-500 or BPC-157 alone, and the reconstitution protocol has to be set by the most fragile component.
Solvent choice
Bacteriostatic water for injection (sterile water with 0.9 percent benzyl alcohol) is the standard solvent for a multi-dose peptide blend. Plain sterile water is acceptable only for single-use experiments because there is no preservative for the multi-week draw window. Normal saline (0.9 percent NaCl) should be avoided. Chloride ions compete for Cu(II) coordination and accelerate transmetalation of the GHK-Cu chelate; the result is a haze, color shift, or visible precipitate within hours. Saline is fine for many single-peptide reconstitutions but it is the wrong solvent for any vial containing a copper peptide.
Why no vortex
The single most important handling rule for the Glow Stack is: do not vortex, do not shake, do not aggressively pipette. Vortexing creates an air-water interface and shear forces that denature peptides via foam and cavitation, and partially dissociates Cu(II) from the GHK chelate. The signature of a damaged blend is a color drift away from the characteristic deep azure-blue toward greenish, teal, or brown. Once that happens the copper has fallen out of the chelate as Cu(OH)2 or hydrolysis products and the GHK-Cu component is no longer in research-usable form. The other two peptides may still be intact, but the blend as a whole has lost the component that gave it its name.
The correct method is the swirl method. Aim the BAC water stream slowly down the inner glass wall of the vial rather than directly onto the lyophilized cake. Swirl or roll the vial between the palms for 30 to 60 seconds until the powder fully dissolves. If undissolved material remains, return the vial to 2 to 8 degrees Celsius for 10 to 15 minutes and swirl again. Patience here protects the most expensive component in the vial.
pH compatibility
The co-stable pH window for the three peptides sits roughly between 5.0 and 6.5. GHK-Cu is most stable in mildly acidic to neutral conditions; above approximately pH 7.4 the Cu(II) chelate begins to release copper and the solution darkens. BPC-157 and TB-500 are both comfortably soluble in the same window. BAC water is naturally near pH 5.5, which is a chemically reasonable solvent for the entire blend without further buffering.
5. IU and dosing math for researchers
U100 insulin syringes calibrate volume in international units where 100 IU equals 1 mL, so 1 IU equals 0.01 mL. With a fixed 70 mg vial, the dose per IU depends entirely on the reconstitution volume the researcher chooses.
Concentration table by reconstitution volume
1 mL BAC water: GHK-Cu 50.0 mg/mL, TB-500 10.0 mg/mL, BPC-157 10.0 mg/mL.
2 mL BAC water: GHK-Cu 25.0 mg/mL, TB-500 5.0 mg/mL, BPC-157 5.0 mg/mL.
3 mL BAC water: GHK-Cu 16.67 mg/mL, TB-500 3.33 mg/mL, BPC-157 3.33 mg/mL.
Per-IU equivalence on a U100 syringe
At 2 mL reconstitution (the typical research middle ground): 1 IU on a U100 syringe corresponds to 250 micrograms of GHK-Cu, 50 micrograms of TB-500, and 50 micrograms of BPC-157. A 20 IU draw therefore delivers 5 mg of GHK-Cu, 1 mg of TB-500, and 1 mg of BPC-157 in the same 0.20 mL injection.
At 1 mL reconstitution: the doses double per IU. 1 IU equals 500 micrograms GHK-Cu, 100 micrograms TB-500, and 100 micrograms BPC-157.
At 3 mL reconstitution: the doses are roughly 167 micrograms GHK-Cu, 33 micrograms TB-500, and 33 micrograms BPC-157 per IU. The trade-off is precision: the smaller the microgram per IU, the more forgiving the syringe granularity, but the larger the injection volume per study event.
Half-life and frequency implications
Vendor-cited research half-lives are roughly 12 hours for GHK-Cu, 7 hours for TB-500, and 4 hours for BPC-157. In a co-administered protocol the three components depopulate at different rates from a single bolus, which is one of the legitimate criticisms of any fixed-ratio blend: the researcher cannot independently titrate frequency for the shorter-half-life components without also re-dosing the longer-half-life one.
6. Storage and stability
Lyophilized vials store at 2 to 8 degrees Celsius protected from light for short to medium term, and at minus 20 degrees Celsius for long-term archival storage of more than 30 days. The lyophilized cake tolerates short ambient shipping windows because all three peptides are stable in the dry state, but the vial should be transferred to refrigeration immediately on arrival.
Once reconstituted in BAC water, the blend keeps for approximately four weeks at 2 to 8 degrees Celsius in an upright vial protected from light. The honest stability ceiling is set by the GHK-Cu component. The other two peptides degrade slowly through aspartate isomerization and glutamine deamidation; the copper chelate degrades faster and more visibly. The first sign of degradation is a color shift from clear pale blue to greenish-brown, followed by haze or visible particulate. That color shift is a discard signal even if the calendar says the vial is still in window.
Three things to never do with a reconstituted blend: never refreeze it, never store it in the refrigerator door (where temperature swings on every door open), and never expose it to direct sunlight. Copper peptides are photosensitive enough that an amber vial or aluminum-foil wrap is a reasonable precaution for any vial that will be drawn from over multiple weeks.
7. Glow Stack vs three separate vials
The blend format is not strictly better than three separate vials. It is a different trade-off. The blend wins on three axes: it consolidates one reconstitution event instead of three (which reduces the probability of an aseptic technique error somewhere in the chain), it locks the three peptides into one ratio so that a single 0.20 mL draw delivers a fixed combination, and it consumes one storage slot in the fridge instead of three.
Three separate vials win on flexibility. A research design that needs to vary the GHK-Cu dose independently of TB-500 cannot do that from the blend without adding a separate GHK-Cu vial anyway. A protocol that needs to compare the combination against each peptide alone (the only honest way to claim synergy) needs the singles in any case. And the shared 4-week reconstitution clock means that if the experiment finishes early, the unused 60 percent of the blend ages out simultaneously across all three components.
The practical heuristic: the Glow Stack is the right format when the ratio is the experimental variable being held constant, and the wrong format when the ratio is the variable being studied. Researchers who want to evaluate a HPLC-verified GHK-Cu, TB-500 and BPC-157 blend as a starting point can do so without forfeiting the option to add singles later in the experimental program.
8. Equipment for subcutaneous research models
The standard equipment for a subcutaneous peptide research workflow is short and self-contained. The blend itself does not change what is needed; it only reduces how many vials are entering the workflow at once.
- U100 insulin syringes, 0.5 mL barrel, 30G or 31G x 8 mm, ideally BD MICRO-FINE+ 0.5 mL U100 insulin syringes for the smallest needle gauge with reliable IU calibration.
- Bacteriostatic water for injection (30 mL multi-dose vial, 0.9 percent benzyl alcohol).
- 70 percent isopropyl alcohol prep pads, one per septum entry and one per injection site.
- A larger reconstitution needle (21 to 23G x 1 inch) plus a 3 mL syringe for the initial BAC water transfer; never reuse this for dosing.
- An FDA-cleared sharps container for single-use needle disposal.
- Powder-free nitrile gloves and a disinfected flat workspace.
- A refrigerator with a verified 2 to 8 degrees Celsius range, ideally with a thermometer or data logger covering the four-week reconstitution window.
For a full kit overview, the injection accessories collection bundles compatible syringes, swabs and sharps containers for research-protocol use.
9. Regulatory framing in the EU and US
None of the three peptides in the Glow Stack are approved drug substances for human therapeutic use in the European Union or the United States. In the EU, supply and possession of unapproved peptides for purposes other than legitimate research can engage the Arzneimittelgesetz (AMG) and equivalent national medicines acts; many vendors ship explicitly under research-use-only terms with the consignee declaring laboratory or institutional use. The German BfArM and equivalent agencies (ANSM in France, AEMPS in Spain, AIFA in Italy, URPL in Poland) treat unauthorized human use of these compounds as outside the scope of the marketing authorization regime entirely.
In the US, the FDA classifies these compounds as research chemicals when sold and labeled for laboratory use and not for human consumption. Both TB-500 and BPC-157 are explicitly listed under WADA Code S0 (non-approved substances) and have been since 2011 and 2019 respectively, which means any athlete in WADA-tested competition who tests positive faces a sanction regardless of intent or formulation.
Practically, this means that a Glow Stack vial in a research workspace is an in vitro and animal-model reagent. It is not, under any framing in any of the jurisdictions above, a clinical product, and any documentation written around it should reflect that.
10. Preclinical evidence base
The peer-reviewed literature for the three peptides is uneven. GHK-Cu has the broadest base, with several decades of dermal, scalp and cosmetic-research publications, and a reasonable foundation in connective-tissue biology. TB-500 (or rather its parent Tb4) has high-profile cardiac and corneal repair publications in Nature and JID, but the synthetic LKKTETQ fragment specifically has thinner independent literature than the full-length protein. BPC-157 has a large publication count concentrated in tendon, gastrointestinal and angiogenesis models, but the publication count is dominated by a single research group, and the field has been slow to produce independent replication of the largest effect sizes.
The honest summary: the three peptides individually sit somewhere between "intriguing preclinical compound with biological plausibility" and "early-stage research chemistry with insufficient human data." The blend is a research convenience, not an evidentiary upgrade. A reader who wants to evaluate the underlying papers can search PubMed for "GHK-Cu", "Thymosin beta 4 wound", "BPC-157 tendon" and "BPC-157 angiogenesis" to find the canonical entry points.
11. Sourcing quality markers
Quality assessment for any research peptide vial reduces to three documents and one in-vial observation:
- HPLC purity certificate: reverse-phase HPLC chromatogram with a single dominant peak per component, integrated area reported, target at or above 99 percent.
- Mass spectrometry identity: ESI-MS or MALDI-TOF confirming theoretical monoisotopic mass for each peptide. GHK-Cu around 340.79 Da, BPC-157 1419.55 Da, TB-500 fragment in the 2.5 to 3.0 kDa range depending on the exact sequence used by the supplier.
- Batch CoA: per-lot certificate listing HPLC purity, mass-spec result, peptide content (commonly 70 to 90 percent by amino acid analysis), water content (Karl Fischer, below 8 percent), and acetate counter-ion content.
- The blue test: after reconstitution the GHK-Cu component should give the entire vial a clear, deep azure-blue color. A pale, greenish, brown or hazy solution is a pre-failure signal even if the paperwork says otherwise.
Vendors that publish lot-matched CoAs and accept third-party analytical re-testing (Janoshik Analytical, Anabolic Lab and similar independent peptide labs) are the practical gold standard in the research-peptide space. Researchers comparing suppliers can cross-reference the CoA lot number printed on the vial label against the corresponding document; mismatches are a hard discard signal. Bergdorf Bioscience peptide blends are sold with batch-traceable HPLC and mass-spectrometry documentation per the published product specification.
12. Frequently asked questions
Can I reconstitute the Glow Stack with normal saline?
No. Saline (0.9 percent NaCl) should be avoided because chloride competes for Cu(II) coordination and destabilizes the GHK-Cu chelate. Bacteriostatic water for injection is the standard solvent for any blend that contains a copper peptide.
Why does the reconstituted blend look blue?
The blue color is the GHK-Cu(II) chelate. A clear, deep azure-blue solution is the expected appearance after a correct reconstitution. A greenish, teal or brown shift indicates that copper has dropped out of the chelate, which is a discard signal.
Can I vortex the vial to speed up dissolution?
No. Vortexing creates shear and foam that denature peptides and dissociate the GHK-Cu chelate. Use the swirl method: aim the BAC water down the vial wall, then swirl or roll the vial between the palms for 30 to 60 seconds.
How long does the reconstituted Glow Stack last in the fridge?
Approximately four weeks at 2 to 8 degrees Celsius, upright, protected from light. The honest stability ceiling is set by the GHK-Cu component; if the color drifts away from azure-blue before the four-week window closes, discard early.
Can I freeze the reconstituted blend to extend shelf life?
No. Freeze-thaw cycles disrupt the copper chelate and shear the peptide bonds. Aliquot only the lyophilized powder if longer storage is required, then reconstitute small aliquots one at a time.
What concentration should I reconstitute at?
2 mL of BAC water is a typical research middle ground, giving 25 mg/mL GHK-Cu and 5 mg/mL each of TB-500 and BPC-157. Smaller volumes give higher mg-per-IU and faster draws; larger volumes give finer dosing granularity at the cost of larger injection volumes.
Why a blend instead of three separate vials?
One reconstitution event reduces aseptic-technique risk, one fridge slot instead of three, and a fixed ratio for protocols where the ratio is held constant. Three separate vials remain better when the experimental design needs to vary the components independently.
Is there a published study showing GHK-Cu, TB-500 and BPC-157 together?
No peer-reviewed study has tested any pairwise combination of the three. The synergy claim is mechanistic, derived from the three peptides acting on different nodes of overlapping pathways (VEGF, NF-kB, ECM remodeling). Any researcher who needs to claim experimental synergy must run the combination against the components in their own model.
What gauge needle should I use?
For subcutaneous research models, a 30G or 31G x 8 mm U100 insulin syringe is the standard. The BD MICRO-FINE+ 0.5 mL is widely available with reliable IU calibration; smaller barrels (0.3 mL) trade total volume for finer markings.
Are TB-500 and BPC-157 banned in WADA-tested sport?
Yes. Both fall under WADA Code S0, the non-approved-substances category. Any athlete in WADA-tested competition who tests positive for either peptide faces a sanction regardless of formulation or intent. The Glow Stack is sold strictly for in vitro and animal-model research.
What happens if I overdose during a research-model administration?
Acute toxicity in animal models is described as low for all three peptides individually, but coadministration data are sparse. Any animal-model protocol should include defined humane endpoints, veterinary oversight, and IACUC or equivalent ethics-committee approval before initiation.
Where can I source the blend with a documented certificate of analysis?
Bergdorf Bioscience supplies the Glow Stack blend with a per-batch HPLC and mass-spectrometry certificate; cross-check the CoA lot number against the vial label on receipt and refuse delivery if the lot numbers do not match.
13. Medical disclaimer
This blog post is for informational purposes only and is not a substitute for professional medical advice. GHK-Cu, TB-500 and BPC-157 are experimental peptides and are not approved for human use in the European Union, the United States, or most other jurisdictions. Use outside of clinical trials or approved indications is illegal in many jurisdictions. TB-500 and BPC-157 are explicitly listed under WADA Code S0 as non-approved substances.
All information regarding mechanisms, reconstitution, dosing math and storage is based on available preclinical research and vendor documentation, but may be inaccurate or incomplete. Individual responses, safety profiles and pharmacokinetic parameters can vary significantly between species and between in vitro and in vivo models. The reconstitution math and per-IU equivalences in this article describe physical concentrations in a vial, not human therapeutic doses.
Before using any peptide in a research model, consult the relevant institutional review board, animal-care committee, and biosafety office. This article does not endorse off-label or illegal use of the compounds discussed. The author and parahealth assume no liability for side effects, injuries, regulatory consequences or adverse events arising from the use of the information discussed in this article.
For research purposes only. Not intended for human consumption. EU country law applies.

Leave a comment
All comments are moderated before being published.
This site is protected by hCaptcha and the hCaptcha Privacy Policy and Terms of Service apply.